Heritage Communities Blog

The Invisible Load… and Why Most Family Caregivers Don’t Stop to See What They’re Carrying
Download our free guide "The Invisible Load" to understand and take stock of the caregiving weight you're carrying. It might help you understand what to do next.
Read Article
The Benefits of Community Life: Why Senior Living Can Enrich the Years Ahead
The holidays remind us how good it feels to gather… to share meals, stories, and simple moments. But for many older adults, those moments become less common over time. As friends move away, routines change, and daily tasks get harder, it’s easy for life to feel quiet and isolating. At Heritage Communities, we believe there’s...
Read Article
How to Celebrate the Holidays With Someone in Senior Living
If you’re starting to consider senior living for a parent, the timing may feel complicated… especially around the holidays. You might wonder: Will the season still feel special? Will we still celebrate the way we always have? Will Mom feel left out? We understand those worries. They’re natural, and they come from love. What we...
Read Article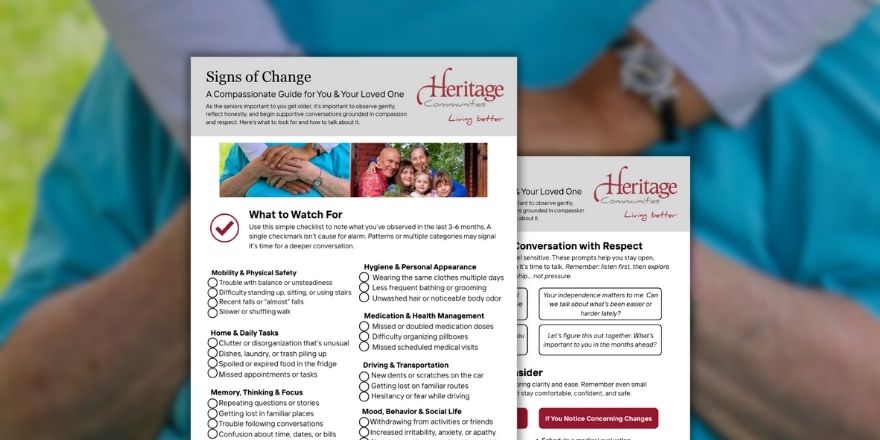
When You Suspect Something’s Changing: A Compassionate Guide for You and Your Parent
If you’re noticing small shifts in your parent’s health, memory, or daily routines, you might be wondering whether you’re overthinking things… or if something really is changing. Download the free PDF checklist to help!
Read Article
Ringing in the Season: Spotlight on Our Bell Choir at Heritage Communities
The holidays are a busy, fun time in senior living! Across our communities, families and groups are getting ready to celebrate with their families and their Heritage family. We’re glad to share a story from Orchard Pointe at Terrazza (a Heritage Community) that really shows what Living Better together looks like. It’s about the wonderful...
Read Article
Does AI Replace Caregivers? The Truth About Technology in Memory Care
In senior living, new AI technology helps caregivers... it doesn't replace them. By handling background monitoring and alerting, AUGi allows caregivers to spend more time where it matters most: engaging with residents, offering comfort, and providing personalized care.
Read Article
How AI Is Redefining Fall Prevention in Memory Care: What Families Need to Know
AUGi, an AI-powered safety companion, is installed in senior living resident apartments and common areas. AUGi uses discreet sensors and predictive analytics to spot fall risks before they become emergencies.
Read Article
Questions to Ask When Touring Senior Living: A Step-by-Step Guide for Families
Choosing the right retirement community is an important, emotional, and complicated decision. But for many families, the big question is: What should I ask when touring a retirement home?
Read Article
Who’s Who In a Senior Living Community: Get to Know Us
Behind every great day at Heritage is someone special making it happen… these are the people you’ll see in our communities!
Read Article
Heritage, Out and About! How Community Outings Help Seniors Live Better
Assisted living isn’t just about care. It’s about helping golden-years adults feel good again! Laughing more, making new friends, and finding purpose in each day. From planned outings into the city to get-togethers in the courtyard, continuing to explore the world is an important part of joining a vibrant community.
Read Article